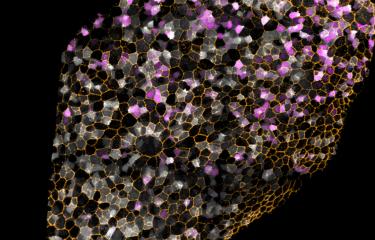
The major challenge currently faced in the field of regenerative medicine is how to reprogram differentiated adult cells into other cell types for tissue repair. Scientists from the Institut Pasteur and the CNRS have made a surprising discovery that cellular senescence, usually associated with aging, cancer and inflammation, promotes reprogramming, where cells revert back to a more embryonic-like state. Converting cells into this "undifferentiated" state represents an important step in developing cell therapy strategies to replace defective cells in pathological conditions, such as disease and aging. These findings are published in the December 22, 2016 issue of the journal Cell Stem Cell.
Scientists have been able to reprogram adult cells to an embryonic-like state, called induced pluripotency, using a set of four transcription factors. This method was established by the Japanese scientist Shinya Yamanaka in 2006, who was awarded the 2012 Nobel Prize in Physiology or Medicine for this discovery. Differentiated adult cells (e.g. cardiac cells, neurons) can thus be reprogrammed to the induced pluripotent state to generate iPS cells[1]. In doing so, they regain the ability to develop into any type of specialized cell. For the past ten years, iPS cells have been a source of high hopes in regenerative medicine and gene therapy, and most notably for modeling diseases, however the differentiation process remains complex and more studies are needed to regulate this process in a controlled manner.
In this study, scientists from the Cellular Plasticity and Disease Modeling Group, directed by Han Li, and the Stem Cells & Development Unit, directed by Shahragim Tajbakhsh (Institut Pasteur/CNRS), set out to investigate the reprogramming of skeletal muscle stem cells in vivo in an attempt to further understand this process and identify factors that could improve it, with the knowledge that muscle is a difficult tissue to reprogram. The scientists observed that "senescent"[2] cells that are produced in response to a muscle lesion in mice, are critical for induced reprogramming of muscle cells via the Yamanaka factors.
They decided to compare the reprogramming rate of muscle cells in the presence and absence of senescent cells. "Surprisingly, the production of iPS cells was more efficient in the presence of senescent cells," explained Aurélie Chiche, lead author of this study. The scientists concluded that senescent cells might enhance the reprogramming process. They then analyzed molecules secreted by the senescent cells and identified Interleukin 6 (IL-6), involved in the acute phase of inflammation, as an important factor to boost reprogramming.
This study discovered an unprecedented role of senescent cells in cellular reprogramming, particularly via IL-6. The challenge now is how to redifferentiate the stem cells obtained this way into specific cell types required for applications in regenerative medicine.
In addition to the support of the institutions mentioned above, this study was supported by LabEx Revive and ERC (European Research Council).
[1] iPS cell: Induced pluripotent stem cell
[2] When a cell experiences stress it ultimately enters a state of senescence, meaning that it ceases to divide. Senescent cells often emerge in hostile contexts such as aging, cancer, irradiation and external aggression.
Source
Injury-induced senescence enables in vivo reprogramming in skeletal muscle, Cell Stem Cell, December 22, 2016
Aurélie Chiche1, 4, Isabelle Le Roux2, 4, Mathieu von Joest1, Hiroshi Sakai2, Sabela Búa Aguín1, Coralie Cazin1, Rana Salam1, Laurence Fiette3, Olinda Alegria1, Patricia Flamant3, Shahragim Tajbakhsh2, *, Han Li1,*,‡
1 Cellular Plasticity & Disease Modelling,
2 Stem Cells & Development,
Dept. of Developmental & Stem Cell Biology, CNRS UMR 3738
3 Human Histopathology and Animal Models
Institut Pasteur, 25 rue du Dr Roux, Paris 75015, France
4 These authors contributed equally and should be considered co-first authors
* Correspondence: Shahragim Tajbakhsh and Han Li
‡ Lead contact